
SPAIN'S National Research Council (CSIC) has announced a new book series seeking to debunk widely-held myths through scientific answers – including whether bread really makes you put on weight.
Sign in/Register
Looking for the Professionals/Advertiser Login?
By Signing up you are agreeing with our Terms and Privacy Policy.Forgot your password?
Feedback is welcome

CLINICAL trials are now well under way with the new 'made-in-Spain' Covid vaccine - 30 participants have been recruited and at least six of them now jabbed in a crucial step closer to a home-grown inoculation being on the shelves.

Hipra Laboratories in Girona (shown above) is developing a formula which uses two different Coronavirus strains, to increase immunity across the board and also to newer, more contagious and more aggressive types.
The first doses have been administered this week at Girona's Josep Trueta Hospital (third picture) and Barcelona's Hospital Clínic, according to Dr Marga Nadal, head of the Girona Biomedical Research Institute (IDIBGI) and Dr Àlex Soriano, head of the infectious diseases unit at Hospital Clínic.
Safety first: Young, healthy volunteers and why everyone is kept in the dark
As a first phase of clinical trials, the aim at the moment is merely to ensure the vaccine is safe, can be tolerated, and 'awakens' the immune system – later phases are concerned with how effective it is, but safety is the initial priority, which is why the jab is being tested on healthy men and women aged at least 18 but under 40, who have not had Covid before and have not yet been vaccinated against it.
Another reason for the young age group is that almost everyone aged 40 and over will now have been inoculated, and the trials have been authorised quickly because any delays would mean an increasingly harder task in finding even 18-plus unvaccinated volunteers.
It was initially reported that the experiment would be 'single-blind', with only the candidates not knowing which vaccine they had been given, but it has since been confirmed the study is a double-blind, meaning the researchers themselves do not know, either.
Records are, obviously, kept, but not knowing which vaccine effects they are dealing with means the researchers eliminate any subconscious temptation to 'look for' or anticipate results they expect to find.
Additionally, as research into drug development is, necessarily in most cases, financed by the laboratories the medication is being created for, there can be no suspicion from any quarters that the investigators were simply telling the fund providers what they want to hear.
Also, with the volunteers not knowing which vaccine they have been given, they will not anticipate certain side-effects and end up reporting psychosomatic ones, or unrelated reactions.
Some have been given the Hipra jab and others have been given a Pfizer, and they will all have a repeat of the same medication after 21 days; this way, nobody will leave the experiment thinking they have been immunised when they have only received a placebo.
Independent authorisation and monitoring
The study has been authorised by the Spanish Medicines and Healthcare Products Agency (AEMPS), the national organisation which has the last word on approving any medication for use or sale, and by Hospital Clínic's Ethics Committee – and also by an independent monitoring committee which keeps a close eye on methodology, clinical practice and on volunteers' response to the injection.

Dr Lorna Leal, specialist at the Clínic's infectious diseases unit and a member of the AIDS and HIV Infection Group at the August Pi i Sunyer Biomedical Research Institute (IDIBAPS) in Barcelona, is heading up the research project.
She says the Hipra vaccine has the advantages of being developed using existing and well-known technology, which means the team was able to use this as a starting point and improve on it.
Based on techniques already in use, the Hipra researchers have been seeking ways of making the vaccine even more safe than those already in use, and capable of inducing a very powerful immune response.
Side-effects are good news, as long as they are bearable and safe
Dr Leal says clinical trials will take a full year to complete, but that so far, those who have been vaccinated already – with the Pfizer or the Hipra, but she and her team do not know which – have nearly all reported 'very mild' side-effects.
Some level of side-effects is always positive news, as it means the immune system is working to fight off the virus by producing antibodies or 'biological weapons' to repel it; no side-effects whatsoever would, therefore, be more of a concern, and this situation is normally greater in the case of vaccine recipients whose existing immune system is less efficient.
Anecdotal evidence so far has shown that younger recipients of the vaccines already in use have tended to suffer greater discomfort than older adults – a sign that the immune response in the young is typically more robust.

But side-effects which could present a greater danger or more discomfort than the condition the vaccine is aimed at preventing are, of course, a negative factor; also, side-effects which the vaccine recipients are unable to tolerate mean more work has to be done to mitigate these.
As an example, an anti-Covid vaccine which commonly caused cancer or heart attacks in recipients would never be approved, as the side-effects would be at least as hazardous to health as the Covid itself; if such a risk existed but was extremely rare, affecting one in 10 million, for example, then it is likely the benefits would be considered to outweigh the level of potential hazard.
If a vaccine was safe but caused weeks of massive headaches, vomiting and diarrhoea, infertility, depression, chronic high blood pressure, hair loss or unbearable all-over itchy skin, then the formula would have to be reworked to include ways of preventing these effects, or given in combination with other pharmaceutical drugs to combat them – although they may not be as dangerous as Covid, nobody can be expected to receive a vaccine which causes these problems, and the financial loss through huge segments of the population being off sick from work would be devastating for the economy.
A few days of 'feeling a bit fluey', a day or two with a mild to moderate headache or nausea and fever – which improves with rest and standard over-the-counter medicines - would be considered acceptable; in fact, these are common responses to the Pfizer vaccine and, although uncomfortable, are very temporary and bearable, and do not affect everyone who receives it.
Why now, when the majority are already immunised?
Given that the vaccine roll-out is likely to be finished by the autumn – at least four regions have now crossed the 70% barrier and most are starting on the 12-19 age group – Hipra has been working on the basis that its formula will be used as an annual booster.
Unlike key pharmaceutical industry countries, such as Germany, The Netherlands and Switzerland, Spain has not had a long history of drug development for humans – in fact, Hipra is a veterinary medication laboratory – meaning funding is not so abundant for these projects and it has taken longer for the Spanish vaccine to get through its various phases of creation.

Hipra has said all along, though, that as other drugs were farther advanced and likely to be on the market much earlier, it was not concerned about being the first in a 'race' to create a preventive medication that would merely contribute to the huge number needed by the world's population over several years.
If nearly 7.5 billion people on earth need to be immunised annually, then the more vaccines available, the better – and greater availability means costs come down, so they are more accessible for less-wealthy countries.
It could, however, be on the market by this time next year, or by 2023, and as Spanish health authorities have taken it as read that the public will need regular boosters, the Hipra contribution will be valuable – and economical, since it will not need to be imported.
Unlike the Pfizer BioNTech jab, one of the first to be approved, it does not present storage issues – whilst the Pfizer has to be kept at exceptionally low temperatures, making it tricky to use in poorer countries which cannot afford the special refrigeration equipment, the Hipra jab is stored at temperatures similar to that of a domestic fridge, or about 2ºC to 8ºC.
What type of vaccine is it?
The Hipra is a PHH-1V formula, along the same lines as the Novavax and the Sanofi/GSK, which are currently being evaluated by the European Medicines Agency (EMA) with a view to being authorised for use in the European Union.
These use one of the proteins in the SARS-CoV-2 virus, the one referred to as the 'S-protein' because of its shape.
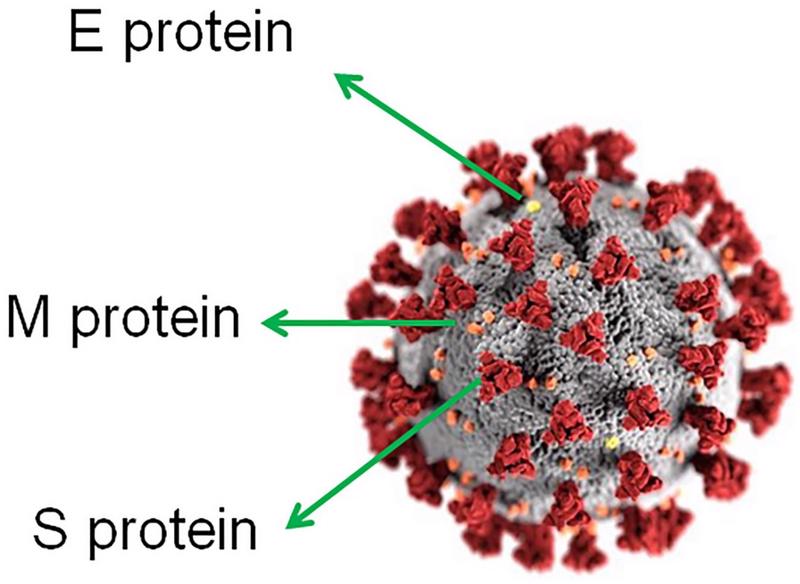
It is named 'S' for 'spike', as opposed to the 'E-protein' ('envelope') and 'M-protein' ('matrix'), as explained by Dr Alissa Eckert and Dr Dan Higgins of the USA's Centre for Disease Control and Prevention (CDC).
The difference between these and the PHH-1V is that the latter will use two 'S-proteins', one from the Alpha and one from the Beta strains – the former originally referred to as the 'British variant', but new mutationes are now known by letters from the Greek alphabet so as not to 'stigmatise' the countries where they first appeared – and which 'lock' together when entering the system.
Hipra researchers hope the two-pronged attack – or four, if each end of the 'S' is considered a 'prong' – will generate an even more potent immune response, and a more 'flexible' and 'versatile' set of antibodies capable of adapting to repel current and future viral mutations, stopping the SARS-CoV-2 from evolving to be able to resist the defensive front mounted by the human organism.

SPAIN'S National Research Council (CSIC) has announced a new book series seeking to debunk widely-held myths through scientific answers – including whether bread really makes you put on weight.

NEW legislation aiming to protect the public from telephone scams and cold-calling is under construction, and will attempt to attack it at source by tightening up on commercial use of customers' personal data.

OUTER space and the Bronze Age do not sit well in the same sentence – they may both have existed at the same time, but anyone based on Earth back then would not have known much, or anything, about what lies beyond.

A FIRM annual fixture for fans of the latest technology, the Barcelona-based Mobile World Congress (MWC) never fails to blow visitors' minds with creations they didn't know they needed. And these cutting-edge...